The Brain & Addiction: How Substance Use Rewires the Brain — and What Recovery Looks Like
Addiction is not a moral failing — it’s a brain disorder that changes the circuits for reward, motivation, memory, and self-control. Those changes make people continue using substances even when the consequences are harmful. This article breaks down the neurobiology behind those shifts and shows how treatment supports the brain’s recovery. You’ll get clear, practical explanations of which brain areas and neurotransmitters are affected, how the addiction cycle rewires circuitry, and which evidence-based therapies and levels of care promote healthy neuroplasticity. Our aim is to turn complex science — from dopamine imbalances to weakened prefrontal control — into straightforward guidance for people getting help, their families, and clinicians planning care. Where helpful, we connect specific interventions, like medical detox and inpatient stabilization, to the neurobiological needs and realistic recovery timelines they address.
Substance use changes both brain structure and function through mechanisms that we can measure — and, importantly, treat. Understanding those mechanisms explains why withdrawal management, behavioral therapies, and ongoing supports are all parts of a complete recovery plan. The sections below cover: structural and functional changes caused by substances; dopamine’s central role in reward and learning; the key brain regions that control decision-making, craving, and stress; how the three-stage addiction cycle maps to neurobiology; and the treatments that help the brain heal. Keywords such as addiction brain changes, neuroscience addiction, dopamine addiction, and brain recovery from addiction are woven through this piece so readers can find focused, evidence-based guidance.
How Does Addiction Affect the Brain’s Structure and Function?
Addiction interferes with the brain’s reward, stress, and executive control systems by changing receptor signaling, synapse strength, and network connections. Over time these neuroadaptations make natural rewards feel less valuable, increase stress reactivity, and weaken top‑down regulation from the prefrontal cortex — all of which contribute to compulsive drug-seeking and poor decision-making. Structurally, we see dendritic changes, altered white-matter integrity, and region-specific volume shifts that reduce cognitive flexibility and impulse control. Clinically, these changes look like intense cravings, risky choices, and a higher chance of relapse, which is why coordinated medical, psychological, and social supports are essential.
Many symptoms flow directly from these neural changes and point to targeted treatment goals such as medical stabilization, practical skill-building, and stress regulation. The next section explains cellular and synaptic mechanisms — like receptor downregulation and tolerance — that underlie these clinical signs.
What Brain Changes Occur During Substance Abuse?
Repeated substance use causes receptor adaptations, tolerance, and synaptic remodeling that alter how neurons communicate and how circuits respond to cues. Drugs can lower receptor sensitivity (downregulation) and change intracellular signaling, so higher doses are needed for the same effect and everyday rewards lose appeal. At the synapse, drug use can strengthen cue–reward associations through long-term potentiation in limbic circuits, reinforcing habitual behavior. These cellular and circuit changes help explain the shift from voluntary use to compulsive patterns — which is why behavioral therapies that retrain learning systems are central to recovery.
With those mechanisms in mind, the next subsection maps the brain areas most affected and the behaviors they drive.
Which Brain Regions Are Most Impacted by Addiction?
Addiction mainly disrupts three interconnected systems: reward circuitry centered on the nucleus accumbens, executive control in the prefrontal cortex, and stress/emotion processing in the amygdala and related limbic areas. The nucleus accumbens assigns value and drives motivation, the prefrontal cortex supports planning and impulse control, and the amygdala heightens negative emotion and stress-triggered relapse. When these regions are out of balance you get the core features of addiction: strong craving, poor decision-making, and increased stress sensitivity. Understanding this map helps explain why successful treatment must both stabilize brain chemistry and rebuild cognitive control and coping skills.
To make these connections concrete, the next section focuses on dopamine — the neuromodulator most closely tied to reward learning and addiction.
What Is the Role of Dopamine in Addiction and the Brain’s Reward System?
Dopamine signals prediction, motivation, and reinforcement learning by marking when outcomes are better or worse than expected. In healthy function, bursts of dopamine signal unexpected rewards or cues that predict them, helping the brain learn what’s worth pursuing. Addictive drugs hijack this system by producing unusually large or prolonged dopamine surges, which speeds up learning for drug-related cues and biases behavior toward substance seeking over other rewards. The result: reduced pleasure from normal activities, stronger cue-triggered cravings, and persistent motivation to use despite the harm.
Different substance classes produce different dopamine patterns and downstream adaptations, which is why withdrawal timelines and treatment needs vary. The table below summarizes typical dopamine effects and withdrawal patterns for common classes of drugs.
How substances change dopamine signaling — and what that means for withdrawal — differs by drug class.
| Substance Class | Typical Dopamine Effect | Typical Withdrawal Timeline |
|---|---|---|
| Stimulants (e.g., methamphetamine, cocaine) | Large, rapid dopamine surges via reuptake inhibition or release | Acute crash in days; prolonged dysphoria and craving weeks–months |
| Opioids (e.g., heroin, fentanyl, prescription opioids) | Indirect dopaminergic increase via mu-opioid receptor activation | Intense physical withdrawal over days; protracted cravings for weeks to months |
| Alcohol | Modulates GABA/glutamate and dopaminergic tone, variable surges | Acute withdrawal 2–7 days (medical risk); protracted mood symptoms weeks to months |
| Sedatives/benzodiazepines | Indirect effects on dopamine through GABAergic systems | Dangerous acute withdrawal risk; extended anxiety and sleep disturbance |
How Does Dopamine Regulate Motivation and Pleasure?
Dopamine signals motivational importance and prediction errors more than raw “pleasure” alone. Phasic dopamine release tells the brain when outcomes differ from expectations, which updates learning and strengthens routines when rewards exceed predictions. With repeated drug use, cues tied to substance use gain strong motivational pull and can trigger craving even when a person consciously wants to stop. That learning-focused role helps explain why people often know the harm but still struggle to resist powerful urges driven by conditioned cues.
That cue-driven learning sets up how different drug classes progressively damage dopamine systems over time — the next section explains those long-term effects.
How Do Addictive Substances Alter Dopamine Mechanisms?
Substances change dopamine function through distinct molecular actions — stimulants rapidly boost extracellular dopamine, opioids disinhibit dopamine neurons, and alcohol alters upstream neurotransmitter balance — but they all reinforce drug-seeking. Acute surges strongly reinforce drug-taking, while chronic exposure leads to compensations like receptor downregulation, lower baseline dopamine tone, and altered synaptic plasticity. Over time these changes cause anhedonia, heightened cue-reactivity, and persistent craving, which maintain the addiction cycle and raise relapse risk. That’s why combining medication where appropriate with behavioral retraining is key to long-term recovery.
The next major section looks at the specific brain regions involved and how their dysfunction shows up in clinical behavior — guiding targeted treatment choices.
What Are the Key Brain Regions Involved in Addiction?
Core regions in the addiction circuitry include the prefrontal cortex (for executive control), the nucleus accumbens (reward valuation), the amygdala (stress and emotion), and the basal ganglia (habit formation). These areas connect via dopamine and glutamate pathways to encode value, form routines, and regulate responses to stress and cues. When any node falters, behavior shifts toward habitual, cue-driven responses and away from flexible, goal-directed control. Clinically, this mapping helps clinicians choose targeted therapies — cognitive skill work for prefrontal deficits, exposure-based strategies for cue reactivity, and stress-reduction practices for limbic overactivity.
Below we look more closely at how the prefrontal cortex supports self-control and how limbic structures drive craving and stress-related relapse, tying brain function to therapeutic targets.
How Does the Prefrontal Cortex Influence Decision-Making and Impulse Control?
The prefrontal cortex (PFC) provides top-down control over impulses, supports planning, and helps evaluate long-term consequences — all skills needed to resist immediate drug rewards. Chronic substance exposure weakens PFC function, causing impaired judgment, poorer working memory, and reduced inhibitory control, which increases relapse risk. Cognitive-behavioral therapies and executive-skill training target these weaknesses through practiced decision-making, problem-solving, and delay-of-gratification exercises. With repeated practice and sustained abstinence, PFC networks can strengthen and real-world self-regulation can improve.
Rebuilding PFC capacity ties directly to neuroplasticity, which we address in later sections on treatments that support structural and functional recovery.
What Roles Do the Nucleus Accumbens and Amygdala Play in Craving and Stress?
The nucleus accumbens (NAc) integrates dopamine and glutamate signals to assign motivational value to cues and actions, making it central to craving and driven drug-seeking. The amygdala encodes emotional salience and stress responses, often increasing negative feelings that drive relapse through negative reinforcement (using to relieve distress). The interaction of NAc cue-salience and amygdala stress reactivity creates powerful relapse triggers, especially under emotional strain. Treatments that lower cue reactivity, build alternative reward pathways, and teach stress-coping skills directly address these limbic mechanisms.
Next, we connect these regional dynamics to the addiction cycle so you can see how brain systems are engaged across stages of use and recovery.
How Does the Addiction Cycle Affect Brain Neurobiology?
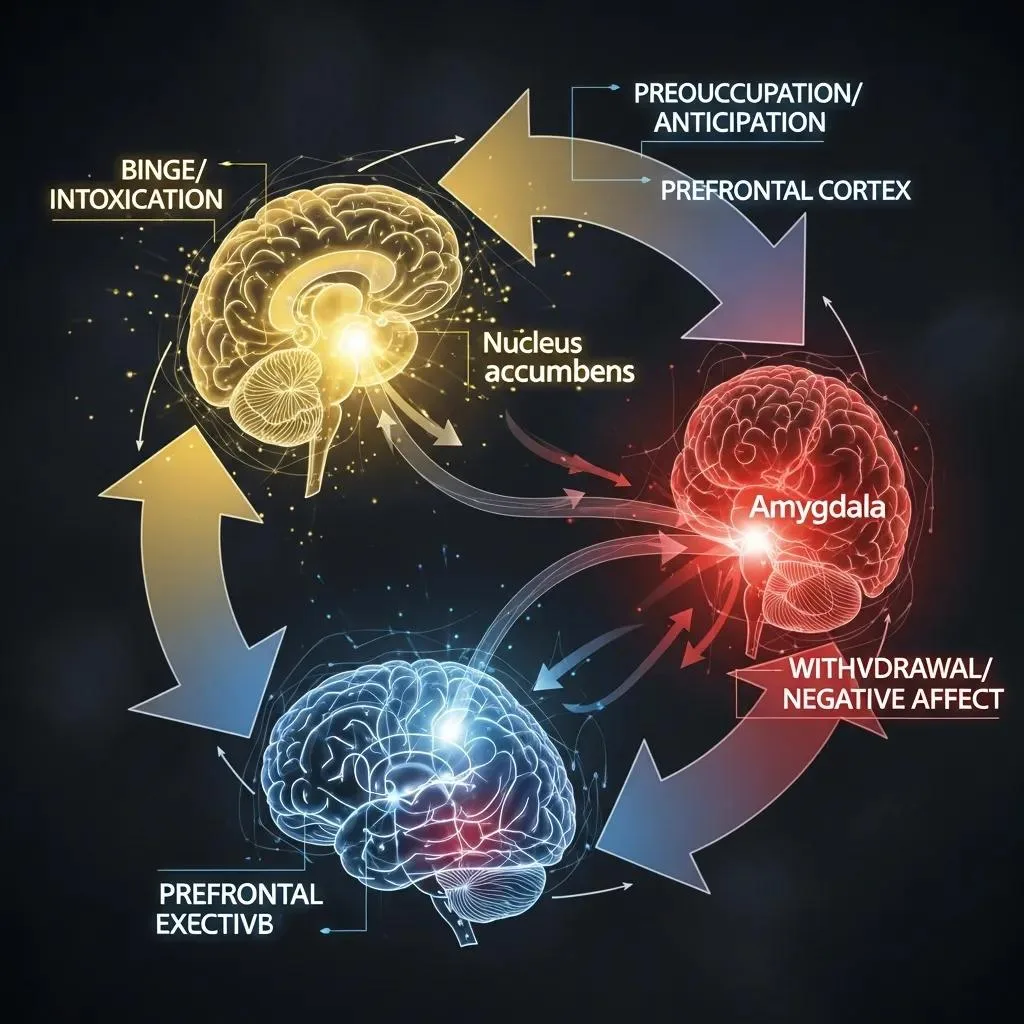
The addiction cycle is typically described in three stages — binge/intoxication, withdrawal/negative affect, and preoccupation/anticipation — each driven by different neural circuits and clinical priorities. Binge/intoxication recruits reward and habit systems (nucleus accumbens, dorsal striatum); withdrawal/negative affect activates stress circuitry (extended amygdala, hypothalamus); and preoccupation/anticipation involves executive control and craving networks (prefrontal cortex and limbic interplay). Mapping stages to brain systems helps match interventions: detox and medical stabilization for withdrawal, inpatient stabilization and intensive therapy for binge/habit disruption, and outpatient relapse-prevention for preoccupation and long-term maintenance.
The table below lays out the three-stage cycle, the primary brain targets, common neurobiological changes, and recommended interventions to make clinical decision-making more transparent.
Here’s a practical mapping of the addiction stages to neural targets and clinical interventions.
| Addiction Stage | Primary Brain Regions | Neurobiological Changes | Recommended Intervention |
|---|---|---|---|
| Binge/Intoxication | Nucleus accumbens, dorsal striatum | Strong dopamine surges, habit formation | Behavioral therapies, inpatient stabilization |
| Withdrawal/Negative Affect | Extended amygdala, hypothalamus | Elevated stress signaling, low dopamine tone | Medical detox, symptom management, inpatient monitoring |
| Preoccupation/Anticipation | Prefrontal cortex, limbic circuits | Cue-induced craving, impaired executive control | Outpatient therapy, CBT, relapse prevention |
Using this stage-to-intervention framework clarifies why matching level of care to the brain’s current state improves safety and outcomes.
In practice, people often step between levels of care as needs change; the next paragraph explains how those care levels respond to stage-related risks.
When deciding next steps, recognizing which stage someone is in helps guide safety and care planning. BetterChoice Treatment Center in Nevada offers pathways aligned with these needs: medical detox to manage withdrawal safely and inpatient rehab for stabilization and intensive therapy. Their multidisciplinary teams and around-the-clock supervision handle medical risk during withdrawal and begin focused work on habit circuitry. Personalized treatment plans, confidentiality safeguards, and coordinated transitions help patients move from acute care toward outpatient relapse-prevention. For Nevada residents seeking timely help, these services connect urgent medical needs with longer-term recovery supports.
Can the Brain Recover from Addiction? What Treatments Support Brain Healing?
Yes — the brain can recover from many addiction-related changes with time, sustained abstinence, and targeted treatments that promote adaptive neuroplasticity. Recovery means reversing maladaptive learned responses, restoring neurochemical balance, and strengthening executive networks so people can respond more flexibly to cues and stress. Evidence-based pathways include medical detox to stabilize physiology, inpatient rehab for concentrated therapy and skills training, outpatient programs for ongoing psychosocial support, and therapies like cognitive-behavioral therapy that retrain decision-making circuits. Pairing these approaches with healthy habits — sleep, exercise, nutrition, and mindfulness — supports synaptic and network-level recovery.
The table below compares levels of care by purpose, typical length, clinical focus, and how each supports neuroplasticity, to help you plan realistic timelines and next steps.
Different levels of care focus on distinct needs, and each contributes to brain recovery in specific ways.
| Level of Care | Purpose | Typical Length | How It Supports Neuroplasticity |
|---|---|---|---|
| Medical Detox | Stabilize withdrawal, ensure safety | Days–two weeks | Restores neurochemical baseline, reduces acute stress signaling |
| Inpatient Rehab | Intensive therapy and stabilization | Weeks (commonly 2–6) | Intensive behavioral retraining and skills practice, high-dose therapy |
| Outpatient Programs | Ongoing therapy and community support | Months–year(s) | Reinforces new behaviors, relapse prevention, gradual network strengthening |
| Counseling (individual/group) | Skill acquisition and trauma work | Variable | Targets executive control, emotional regulation, and cue extinction |
Viewed together, these levels form a stepwise plan that gradually restores healthy network function and lowers relapse risk.
Treatment planning also includes practical intake steps, insurance questions, and clear expectations about timelines and privacy — information that helps families and patients move from recognition to care.
How Do Evidence-Based Therapies Promote Neuroplasticity?
Therapies with strong evidence — such as cognitive-behavioral therapy (CBT), contingency management, trauma-informed care, and medication-assisted treatment when appropriate — each act on neural mechanisms that drive addiction. CBT strengthens prefrontal networks by teaching problem-solving, cognitive reframing, and behavioral skills that reduce impulsive responses. Medication-assisted treatments stabilize chemistry in opioid and alcohol use disorders, lowering craving so people can engage in therapy more effectively. Repeated therapeutic practice and behavioral rehearsal produce synaptic changes that improve circuit function and support lasting change.
These clinical approaches work best when paired with social supports and relapse-prevention planning; the following subsection covers lifestyle steps that further support brain health during recovery.
What Holistic Approaches Enhance Brain Health During Recovery?
Healthy lifestyle habits — consistent sleep, balanced nutrition, regular exercise, mindfulness, and supportive relationships — complement formal treatment by lowering inflammation, supporting neurogenesis, and improving mood regulation. Exercise increases brain-derived neurotrophic factor (BDNF) and supports hippocampal health; sleep repairs cognitive and emotional systems; and mindfulness reduces limbic overactivity that fuels craving and stress reactivity. Practical steps include setting a sleep routine, adding gradual aerobic activity, and using simple breathing or mindfulness practices to manage cravings. These lifestyle elements speed the adaptive neuroplastic changes initiated by therapy and help sustain recovery over time.
For people in Nevada planning care, embedding these supports into a structured treatment pathway — starting with safe detox and moving through inpatient or outpatient programming — creates a comprehensive route toward brain healing. BetterChoice Treatment Center coordinates medical detox and inpatient rehab with personalized plans and holistic supports, while emphasizing confidentiality, multidisciplinary care, and continuous supervision during high‑risk periods. Families can ask about intake steps, expected timelines, and how insurance may apply when exploring local options.
- Intake Steps: Call to describe substance use and any medical risks, complete a clinical screening, and determine the appropriate level of care.
- Typical Timelines: Expect medical detox to last days to two weeks, inpatient programs commonly two to six weeks, and outpatient care to continue for months as relapse-prevention work continues.
- Privacy and Safety: Ask providers about confidentiality, 24/7 clinical coverage, and multidisciplinary supports for co-occurring medical or psychiatric needs.
- If you live in Nevada and need help, call the provider or use local intake options to request an initial screening and safety assessment.
- For families, prepare a short history of the person’s substance use, medical concerns, and recent treatment attempts to speed triage.
- Clinicians coordinating care should align detox, inpatient, and outpatient transitions with neurobiological stage and symptom severity to lower early relapse risk.
- If immediate medical risk is present: Prioritize medical detox with 24/7 supervision to manage withdrawal safely.
- If motivation is high but unstable: Inpatient rehab gives concentrated therapy to rebuild coping skills and reduce cue-reactivity.
- If stability exists: Outpatient therapy and ongoing supports can maintain gains and strengthen executive control networks.
These options form a practical roadmap tied to neurobiological needs and recovery timelines, keeping the focus on safety, evidence-based care, and long-term brain healing.
Frequently Asked Questions
1. What are the long-term effects of addiction on brain health?
Long-term substance use can change brain structure and function — for example, reduced gray matter volume, poorer cognitive flexibility, and disrupted neurotransmitter systems. Those changes may lead to memory problems, emotional instability, and increased stress sensitivity, which can make decision-making and resisting cravings harder and raise relapse risk. The good news: many of these effects can improve with appropriate treatment and sustained support.
2. How can family members support a loved one in recovery?
Families are vital to recovery. Offer steady emotional support, encourage participation in treatment, and help create a stable, healthy environment. Communicate without judgment, listen actively, and educate yourselves about addiction and recovery so you can provide informed, practical help. Family engagement often improves outcomes and helps sustain long-term recovery.
3. What role does stress play in addiction and recovery?
Stress is a major driver of both the development of addiction and relapse risk. High stress can trigger cravings and increase the likelihood of returning to use. Effective stress management — including therapy, exercise, mindfulness, and healthy routines — strengthens resilience and reduces vulnerability to triggers, supporting better recovery outcomes.
4. Are there specific therapies that are more effective for certain types of addiction?
Yes. CBT is broadly effective across many substance use disorders because it teaches skills to change thoughts and behaviors. Medication-assisted treatment (MAT) is especially effective for opioid and alcohol use disorders by stabilizing brain chemistry and reducing cravings while patients engage in therapy. Matching therapy to the substance and the person’s needs improves treatment success.
5. How does neuroplasticity contribute to recovery from addiction?
Neuroplasticity is the brain’s ability to form new connections and reorganize itself. In recovery, neuroplasticity lets the brain undo some of the maladaptive learning caused by substance use and build healthier circuits through therapy, practice, and supportive routines. Over time, those new patterns help reduce cravings, improve decision-making, and support sustained recovery.
6. What lifestyle changes can support brain recovery during addiction treatment?
Healthy habits boost brain recovery. Regular aerobic exercise raises BDNF and supports cognitive health; a balanced diet nourishes brain function; consistent sleep restores emotional regulation; and mindfulness lowers stress reactivity. Combined with formal treatment, these changes speed recovery and improve long-term resilience.
7. How can individuals prevent relapse after completing treatment?
Preventing relapse requires ongoing effort and systems of support. Build a robust support network of friends, family, and peer groups. Engage in aftercare like outpatient therapy or sober living when appropriate. Identify triggers, practice coping strategies, and maintain routines that support mental and physical health. Setting realistic goals and staying connected to treatment resources strengthens long-term recovery.
Conclusion
Understanding how addiction changes the brain clarifies why targeted, evidence-based care works: it stabilizes neurochemistry, retrains learning systems, and rebuilds executive control. A comprehensive treatment plan — from medical detox to ongoing therapy and lifestyle supports — gives the brain what it needs to heal. If you or a loved one is ready to take the next step, explore the resources at BetterChoice Treatment Center to begin a guided, compassionate path toward recovery.
